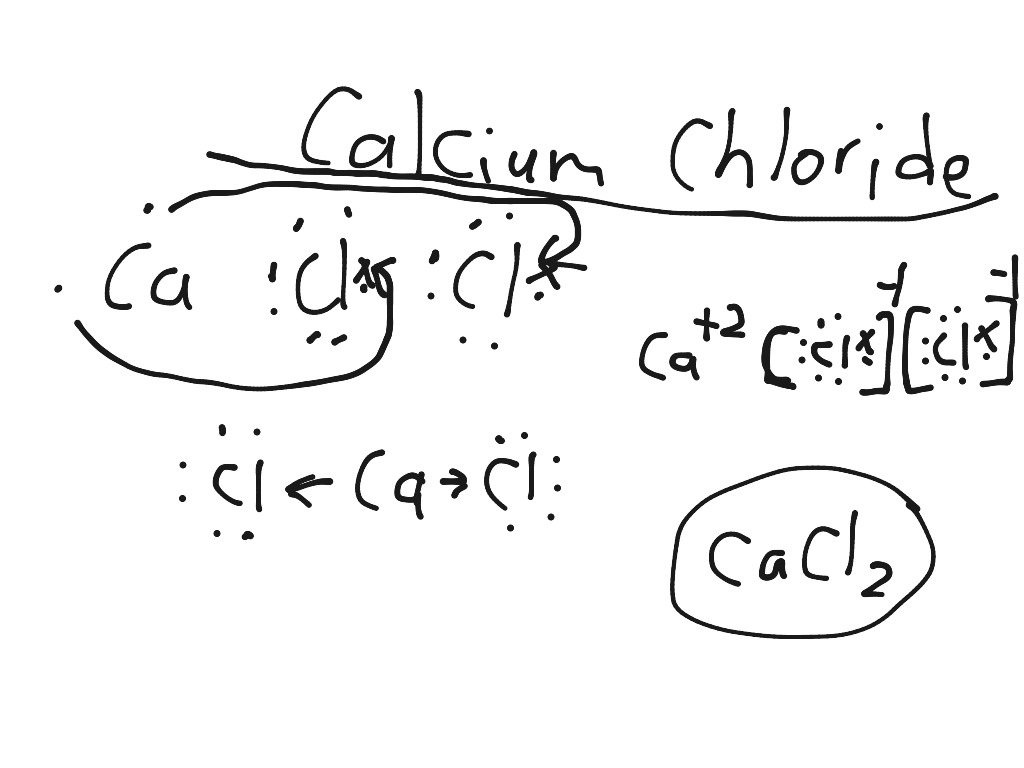
Nanoscale elemental powders and suspensions, as alternative high surface area forms, may be considered.Į509 (Calcium chloride) (CaCl₂) is one of the most versatile chemicals with endless applications. Ultra high purity and high purity compositions improve both optical quality and usefulness as scientific standards. They can form either inorganic or organic compounds.Į509 (Calcium chloride) is generally immediately available in most volumes. The chloride ion controls fluid equilibrium and pH levels in metabolic systems. 
Ultra high purity and proprietary formulations can be prepared. They are formed through various chlorination processes whereby at least one chlorine anion (Cl-) is covalently bonded to the relevant metal or cation. Impurities such as alkali chlorides, magnesium, and calcium hydroxide should be within specified composition limits.Į509 (Calcium chloride) is an excellent water soluble crystalline Calcium source for uses compatible with chlorides.Ĭhloride compounds can conduct electricity when fused or dissolved in water.Ĭhloride materials can be decomposed by electrolysis to chlorine gas and the metal. Two types of E509 (Calcium chloride) are covered, as follows: Type S (solid), flake, pellet, or granular E509 (Calcium chloride) in varying concentrations and Type L (liquid), water solutions of E509 (Calcium chloride) in varying concentrations.Ĭoncentrations shall be expressed as a percentage of the total. Hence, when used as a food additive, E509 (Calcium chloride) does not pose the same health risks (specifically, high blood pressure) that exist with sodium-based salt solutions. This inorganic compound is a colorless, crystalline solid that is highly soluble in water.Īlthough similar to table salt in composition, the compound contains calcium instead of sodium. 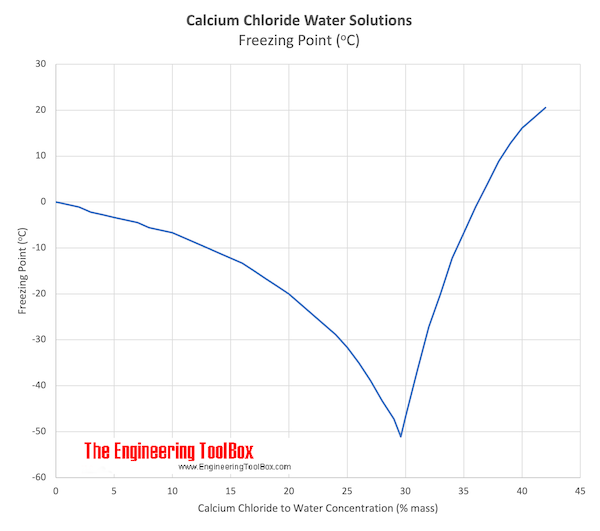
The compound can be useful for displacing ions from solution for example, phosphate may be displaced from solution by calcium.Į509 (Calcium chloride) is a form of salt - but not the type you sprinkle on your food. Sinks and mixes with water.Į509 (Calcium chloride) is an inorganic compound, a salt with the chemical formula CaCl2.Į509 (Calcium chloride) is a white colored crystalline solid at room temperature, and it is highly soluble in water.Į509 (Calcium chloride) can be created by neutralising hydrochloric acid with calcium hydroxide.Į509 (Calcium chloride) is commonly encountered as a hydrated solid with generic formula CaCl2(H2O)x, where x = 0, 1, 2, 4, and 6.Į509 (Calcium chloride) is mainly used for de-icing and dust control.īecause the anhydrous salt is hygroscopic, E509 (Calcium chloride) is used as a desiccant.Į509 (Calcium chloride) is a white deliquescent salt CaCl2 used in its anhydrous state as a drying and dehumidifying agent and in a hydrated state for controlling dust and ice on roads.Į509 (Calcium chloride) is a naturally occurring salt derived from limestone.Į509 (Calcium chloride) is a white solid and can also be produced synthetically.Į509 (Calcium chloride) is solid at room temperature and dissolves in water.Į509 (Calcium chloride) is an ionic compound of calcium and chlorine with the chemical formula CaCl2 and CAS 1.Īt room temperature, it is a colorless crystalline solid that is highly soluble in water.Į509 (Calcium chloride) is used in industry as a de-icer, in road surfacing to suppress the formation of dust, in water treatment to increase the hardness of water in swimming pools, and as an additive in plastics and fire extinguishers.Į509 (Calcium chloride), Dihydrate is a white deliquescent compound which is commonly used as a reagent in biochemistry.Ĭalcium plays important roles in many biological processes, including signal transduction, muscle contraction, maintenance of cell membranes and cell wall stability.Į509 (Calcium chloride) is a soluble compound that can serve as a source of calcium ions in a solution. Physical Description of E509 (Calcium chloride):Į509 (Calcium chloride) is a white to off-white solid. 
E509 (Calcium chloride) = Calcium dichloride = CaCl2 = Calciumchloride
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
